Receive the latest news, event invites, funding opportunities and more from the Ontario Institute for Cancer Research.

Cancer Therapeutics Innovation Pipeline (CTIP) supports the local translation of Ontario discoveries into therapies with the potential for improving the lives of cancer patients while creating a pipeline of promising drugs to attract partnerships and investment to Ontario.
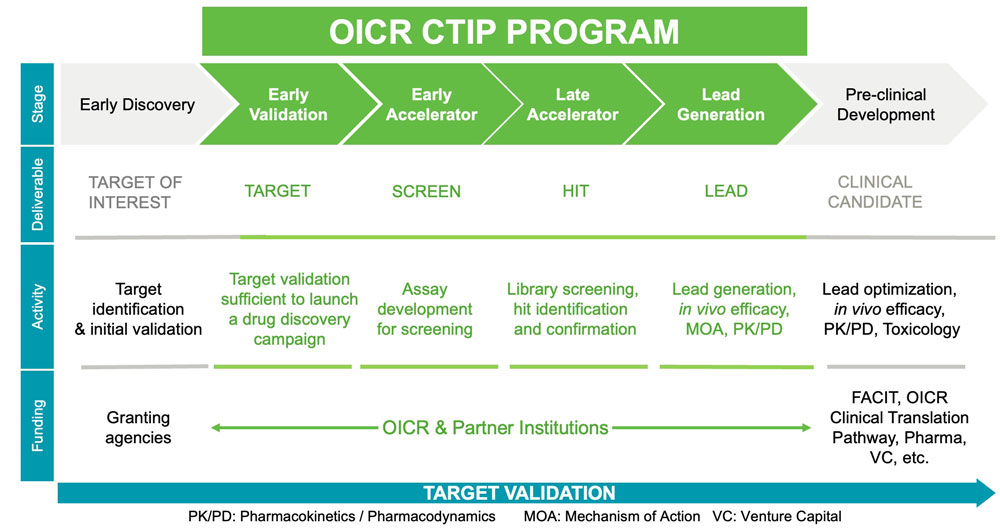
Figure 1: The Cancer Therapeutics Innovation Pipeline: Stages, deliverables, major activities, and funding sources.
CTIP funds projects in four stages of preclinical drug discovery as shown in Figure 1:














Hakim Djaballah
Founder, President and CEO, Keren Therapeutics
TPAC Chair | TPAC member since 2017

Vahe Bedian
Co-founder and Executive Advisor, Viridian Therapeutics
TPAC member from 2017 – 2021 and 2025 – Present

Alexander Bishop
Director, Center for Childhood Cancer Research, Nationwide Children’s Hospital
Professor of Pediatrics, The Ohio State University
TPAC member since 2026

Elizabeth Eisenhauer
Professor Emerita, Queen’s University
TPAC member since 2017

Marc Ferrer
Director, 3-D Tissue Bioprinting Laboratory, National Center for Advancing Translational Sciences
TPAC member since 2020

Terry Hawrysh
Patient partner
TPAC member since 2026

Zaneta Nikolovska-Coleska
Associate Dean, Graduate & Postdoctoral Studies
University of Michigan Medical School
TPAC member since 2021

Ruth Plummer
Professor of Experimental Cancer Medicine
Newcastle University
TPAC member since 2025

Attila Seyhan
Director of Translational Oncology Operations, Brown University
TPAC member since 2020
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |